Does More Calcium Mean More Kidney Stones? The Chemistry Explained
Calcium is one of the major minerals essential for human health. Apart from being a key component of bones and teeth, it is required for blood clotting, transmission of nerve impulses, muscle contraction and cell metabolism1. Calcium also helps delay bone loss with aging, reducing the risk of osteoporosis. For most adults, the recommended dietary allowance (RDA)2 is 1,000 mg/day, increasing to 1,200 mg/day for women >50 and men >70 years old.
However, a study conducted by Food and Environmental Hygiene Department in 2014 revealed that more than 90% of Hong Kong adults consumed less than the recommended 1,000 mg of calcium daily, with the average intake at only 430mg3! Still, many people are concerned that calcium intake, especially from calcium rich food or health supplements, might increase risk of kidney stone formation.
Is it true that the more calcium you consume, the higher your chance of developing kidney stones? This article clarifies this question by examining the chemistry behind calcium and kidney stone formation.
What is a Kidney Stone?
Kidney stones are mineral deposits that form in the urinary system. They develop when increased supersaturation of urine causes the dissolved salts and minerals to precipitate. There are different types of kidney stones, with 80 - 90% composed of calcium stones in the form of either calcium oxalate or calcium phosphate. Calcium oxalate is the most common type, accounting for around 75% of all kidney stones4. Uric acid stone (5-10%) is another common type and form when uric acid - a waste product in the body - crystalizes in acidic urine. Less frequently, there are struvite stones that are related to chronic urinary tract infections, and cystine stones which is developed due to a rare metabolic disorder5. Low urine output and dehydration are major risk factors for all types of kidney stones. For calcium stones specifically, hypercalciuria, hyperoxaluria, and hypocitraturia are the primary risk factors. Among these, hyperoxaluria is the leading cause of the most common calcium oxalate monohydrate stones5. A local survey estimated the household point prevalence of kidney stones was 6.9% in Hong Kong6.
Once formed, the kidney stones may remain in the kidney or travel down the urinary tract to the ureter or bladder (as shown in Figure 1). Small stones can be excreted in urine, but larger stones may become lodged within a ureter, causing symptoms such as severe lower back pain, blood in the urine, nausea, vomiting, fever and chills, or foul-smelling urine.
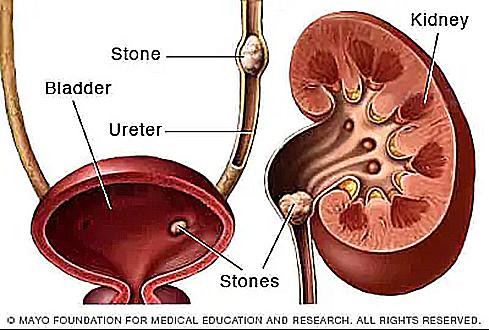
Figure 1: Locations of kidney stones inside the renal system
Can a Calcium Restricted Diet prevent Kidney Stone formation?
Since calcium oxalate is the predominant type of kidney stone, most research has focused on its prevention and management. Historically, the key treatment strategy for patients with calcium urinary stones or recurrent kidney stone was a calcium-restricted diet. It was believed that reducing dietary calcium would limit the amount excreted in urine, thereby decreasing the risk of calcium stone formation. However, large scale studies including Health Professionals Follow-up Study and Nurses Health Studies I and II demonstrated that low dietary calcium intake actually increases the risk of calcium oxalate stones formation7. Conversely, recent large prospective observational studies in men and women consistently support a reduced risk of stone formation with increased dietary calcium intake8.
Role of Calcium in Kidney Stone formation
Calcium is absorbed in ionic state in the gastrointestinal tract. When calcium either from dietary or supplemental sources is consumed with oxalate rich food, an insoluble calcium oxalate complex forms in the intestinal tract and is excreted in the stool, limiting intestinal absorption of oxalate. This results in decreased urinary excretion of free oxalate. However, a restricted calcium diet increases the amount of free oxalate for intestinal absorption, leading to greater urinary excretion of oxalate9. Since calcium is typically present in relative excess in the urine, urinary oxalate is considered more significant than a large increase in urinary calcium.
Chemical reactions related to formation of Calcium oxalate in urine4
CaCl2 (aq) → Ca2+ (aq) + 2Cl- (aq) Calcium ions come from dietary sources and bone resorption
C2O4H2(aq) → C2O42- (aq) + 2H+ (aq) Oxalate ions come from dietary sources and metabolic processes
Ca2+ (aq) + C2O42- (aq) ⇋ CaC2O4 (s) ↓ Formation of calcium oxalate crystals
The formation of calcium oxalate crystals is primarily driven by the urinary supersaturation of calcium and oxalate ions, resulting in precipitation when concentrations exceed solubility. Low urine pH slightly favors calcium oxalate precipitation, but not as dramatically as for other stone types like uric acid crystals.
Multiple studies have confirmed the negative corelation between low calcium intake and increase urinary oxalate excretion. Using the standardized [13C2] oxalate absorption test, it has been shown that oxalate absorption decreases across a range of 200 to 1,800mg daily calcium intake9 as depicted in Figure 2.
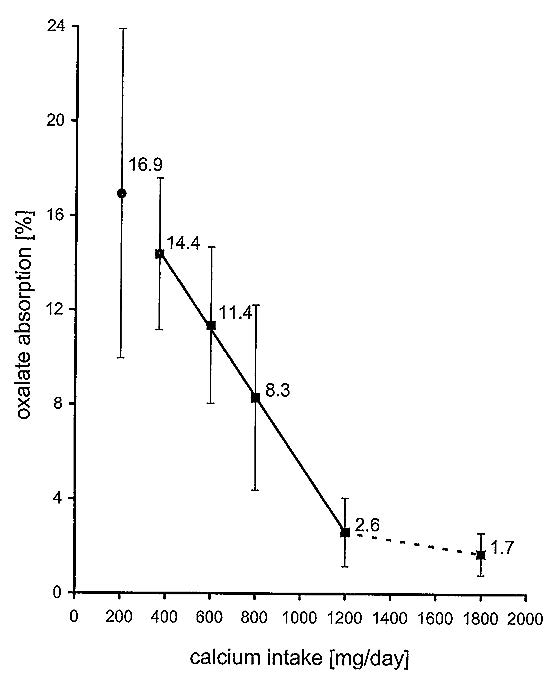
Figure 2: Mean oxalate absorption values at 200 mg Ca/day up to 1800 mg Ca/day. For the range of physiologic Ca intakes, 370 to 1200 mg Ca/day, the graph was a straight line with an r of -0.9997. The dotted connecting line from Ca intakes of 1200 to 1800 mg/day indicates the reduced slope - i.e. the reduced efficacy of additional Ca.
There is a marked change in the slope of the oxalate absorption curve when calcium intake exceeds 1,200mg daily. Additional calcium either from diet or supplements added to a low-calcium diet (e.g., 400mg daily) can lower the risk of calcium oxalate crystal formation. This is because the decrease of oxalate absorption and resulting decrease in oxalate excretion overcompensates the increase in calcium absorption. However, calcium supplementation to a high-calcium diet (1,200mg or above) only marginally reduces oxalate absorption, without offsetting the increased calcium excretion9.
The dependence of oxalate absorption on calcium intake explains why a low-calcium diet increases the risk of kidney stone formation, as observed in various clinical and epidemiological studies.
Dietary Advice to Prevent Kidney Stones5
- Drink enough fluid to keep the urine less concentrated with waste materials.
- Consume adequate amount of calcium from food within 1,000 – 1,200 mg/day. If calcium supplement is required, it is recommended to take it with meals7.
- Restrict salt intake - high salt content will reduce calcium reabsorption from urine to blood, increasing kidney stone risk.
- Limit intake of animal protein – high animal protein increases urine acidity, facilitating formation of calcium oxalate and uric acid stones.
- Eat food high in fiber such as fruits and vegetables.
- Limit oxalate-rich foods (e.g., spinach, chocolate, nuts) for those prone to calcium oxalate stones or recurrent stones formation.
Conclusion
There is strong evidence that adequate dietary calcium intake is protective against calcium stone formation. In fact, a calcium-restricted diet may decrease bone density or increase osteoporosis risk in addition to raising the risk of calcium kidney stones. Many people especially those with a history of kidney stones, mistakenly believe that reducing dietary calcium is beneficial. More public education is needed to emphasize that sufficient calcium intake can actually prevent calcium stone formation.
However, individuals with hypercalciuria, certain metabolic disorders, or those taking specific medications should consult healthcare professionals regarding their calcium intake. For most people, maintaining an adequate dietary calcium intake is recommended, as it is essential for overall health and helpful in preventing kidney stones.
References
- Byrd-Bredbenner, Carol. et al. (2019) Wardlaw's Perspectives in Nutrition. McGraw Hill Education. 11th Ed.
- National Institute of Health Office of Dietary Supplements
https://ods.od.nih.gov/factsheets/Calcium-HealthProfessional/ - The First Hong Kong Total Diet Study: Minerals. (2014, December). Center for Food Safety, Food and Environmental Hygiene Department, HKSAR.
- Nazarian R., et al. What causes calcium oxalate kidney stones to form? An update on recent advances. Uro 2025,5,6.
https://doi.org/10.3390/uro5010006 - Peerapen P. and Thongboonkerd V. Kidney Stone Prevention. Advances in Nutrition 14 (2023) 555 - 569
- Chan WH Steve, et al. A report on a randomly sampled questionnaire survey about renal stone disease in Hong Kong. Hong Kong Med J 2008;14:427-31
- Sorensen Mathew D. Calcium intake and urinary stone disease. Translational Andrology and Urology. 2014;3(3):235-240
- Taylor EN and Curhan GC. Diet and fluid prescription in stone disease. Kidney International (2006) 70, 835–839. doi:10.1038/sj.ki.5001656
- Von Unruh Gerd E., et al. Dependence of Oxalate Absorption on the Daily Calcium Intake. Journal of the American Society of Nephrology 15: 1567-1573, 2004.
By K.L. Tang, Nutritionist, HKBU chemistry alumnus